Electronic Data Capture for Clinical registries
Clinical research - Registry - Data Capture - Analysis
The use of clinical data management systems has become essential in clinical trials to handle large amounts and various types of data. At Swiss4ward we offer our Clinical Data Management System (CDMS) software called CRF.ONE. It is a cloud-based clinical data management system, enabling researchers/investigators & patients to capture and manage clinical data. The data security is being followed according to the GDPR and Good Clinical Practice (GCP) guidelines.
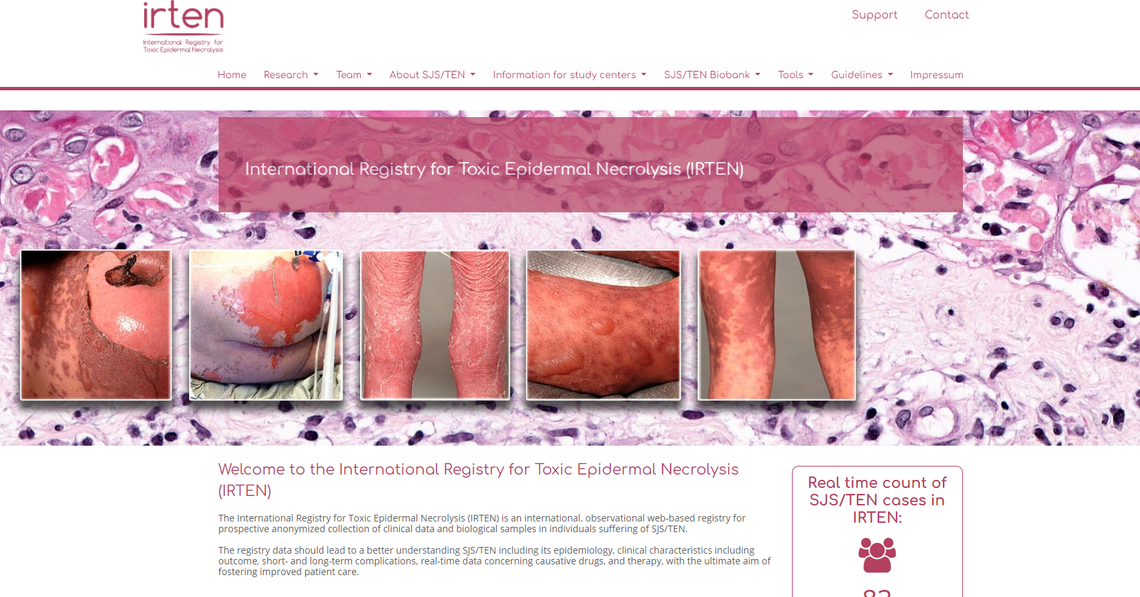
We have experience providing clinical research solutions for registries such as SDNTT, IRTEN, HSBest, HSR-CH, ADBest-TREAT and many more.